光学生物传感器
综合亲和力和动力学
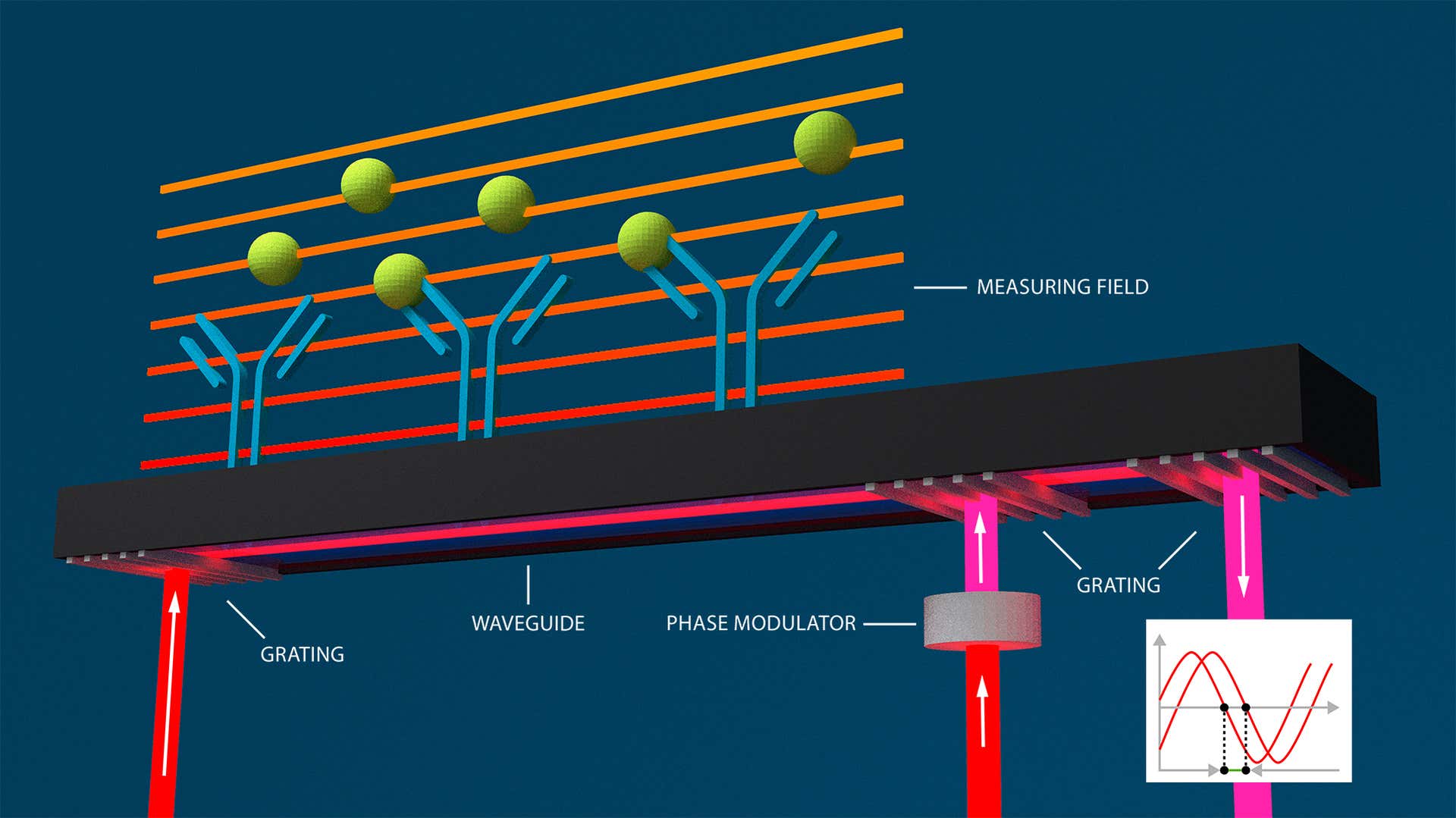
综合亲和力和动力学

从本质上讲,传感器是一种测量物理变化并将其转换为可检测输出(例如电流变化)的设备。让我们通过一些简单的例子来探索什么是传感器和生物传感器。
变化可能发生在环境中。一个很好的例子是冰箱里的温度传感器。随着温度的升高,电阻发生变化。这意味着在温度过高时,传感器会让电流通过并激活冷却系统。
当传感器暴露在样品中时,这种变化也会发生。例如 pH 计,它可以测量浸入样品时电流的变化,并读出样品的 pH 值。在生物传感器中,传感组件属于生物学来源(例如抗体),且可以感知变化(例如抗原的结合),然后这种变化会被转换成可检测的输出(例如光源强度的变化)。
许多光学生物传感器具有微流控,这意味着缓冲液和样品的流动在系统中交替进行。在实验中,其中一种相互作用物固定在传感器表面(配体),另一种相互作用物在通过传感器表面的溶液(分析物)中自由存在。
具有微流控的光学生物传感器及其灵敏,且可以通过无标记定量测量生物物理的相互作用。非无标记的技术需要对组件进行标记方可获得可检测到的输出(例如,荧光标记)。这些标记技术利用标记同位素、荧光标记和放射性标记,这些标记往往会改变物理化学或结合性质,让准确地分析其相互作用变得困难。而且,标记可能会改变影响实验结果的配体的结构和/或功能。此外,光学生物传感器实时测量相互作用,提供相互作用动力学的准确数据。ELISA 分析等技术只能提供端点测量。
光学生物传感器可以提供实时、无标记的综合亲和力和动力学。分析人员可以直接测量分子之间的实时相互作用,包括结合、平衡和解离的每个阶段,这为相互作用的亲和性和动力学提供了更深入的见解。从药品发明到研究,结合动力学与许多应用相关,应用范围包括:

下一代生物分析平台
基于专有的 GCI 技术构建而成,分析人员可以直接测量分子之间的实时相互作用,包括结合、平衡和解离的每个阶段,这为相互作用的亲和性和动力学提供了更深入的了解。从药品发明到研究,结合动力学与许多应用相关,应用范围包括 WAVEcontrol 和 waveRAPID 软件,在保持通量的同时实现卓越的数据分析。
同时,WAVEchip®,我们的无堵塞微流试剂盒,支持广泛的样品类型和尺寸。WAVEsystem 是一种 Creoptix 技术,配有一个温控自动进样器,可安装小瓶、两个 96 孔板或一个 384 孔板,以进一步方便使用。总之,该系统可以为您提供一个卓越的实时无标记结合动力学平台: